-

What Pascuala Stands For
At Pascuala Distribution Trading Corporation, we distribute trust — not merely as a tagline, but as a standard that defines how we select, represent, and deliver every product. That trust is earned through science, consistency, and the confidence we instill in the clients who rely on our solutions.

SAFETY
Trust begins with safety. Every product in our catalog is carefully screened for compliance with international health regulations and Philippine safety standards. We work exclusively with manufacturers that hold KFDA, CE, ISO, and Philippine FDA certifications — ensuring each formulation is clinically safe and rigorously tested.

CREDIBILITY
We represent what’s proven. Pascuala collaborates only with globally recognized brands and laboratories, offering our partner physicians, dermatologists, and certified practitioners products they can confidently use in their practice. Every solution we carry reflects our commitment to clinical integrity and professional credibility.

PERFORMANCE
We deliver results that can be seen and felt. Whether used in aesthetic procedures, skin regeneration protocols, or non-invasive rejuvenation treatments, our products are built for visible transformation and lasting improvement. It’s not just about beauty — it’s about performance that supports your expertise and strengthens client trust.
-

Next-Generation Gold-Coated Ethosome Technology for High-Efficacy Aesthetic Delivery
EthosomePTT is an upgraded, micro-particle solution derived from ethosome, a lipid-based delivery vesicle known for its deep skin penetration. Compared to traditional liposomes, ethosomes are 3–4 times more effective at delivering active ingredients into the dermis and sub-dermal layers. The “PTT” upgrade refers to the coating of the ethosomes with colloidal gold and platinum, forming a multi-layered particle system that increases both stability and delivery efficiency.
This upgraded structure acts as a shield against degradation caused by light or heat and provides a negatively charged surface, improving compatibility and attraction to the skin barrier.Scientific Background: From Liposome to Gold-Coated Ethosome
FEATURE LIPOSOME (1960s) Ethosome (2000) Gold-Coated Ethosome (2021) Skin Permeability Low Moderate-High Very High Electric Charge None Negative Negative Light/Heat Stability Low Moderate High Layer Complexity Single-layer Bi-layered Multi-layered with metals EthosomePTT’s development was built upon the 2000 innovation by Prof. E. Touitou, who first proposed ethosome as a solution to overcome the limitations of liposomes. In 2021, N-Finders advanced this further by incorporating noble metals—gold and platinum—into the ethosomal structure, increasing transdermal permeability, ingredient stability, and controlled release.
PRODUCT VARIANTS
EthosomePTT Brightening Solution
Designed to target melanin inhibition, dullness, and uneven tone, this brightening solution is formulated with:
- Niacinamide
- Tranexamic Acid
- Ascorbic Acid
- Ethyl Ascorbyl Ether
- Centella Asiatica Extract
- Colloidal Gold and Platinum
- Glutathione, Trehalose, and Hyaluronic Acid
These ingredients are delivered deep into the skin layers via the enhanced ethosomal system, providing intensive brightening without triggering inflammation or irritation.
EthosomePTT Anti-AC Solution
Tailored for acne-prone and inflamed skin, the Anti-AC solution contains:
- Niacinamide
- Centella Asiatica Extract
- Salicylic Acid
- Tranexamic Acid
- Ascorbic Derivatives
- Colloidal Gold and Platinum
- Betaine and Methylpropanediol
This formulation controls sebum, calms the skin, and supports skin barrier regeneration while penetrating effectively into clogged follicles.
KEY BENEFITS
- Multi-Layered Penetration
- Gold- and platinum-coated vesicles enhance both depth and consistency of transdermal absorption.
- High Ingredient Stability
- Prevents ingredient oxidation or degradation even when exposed to light or temperature fluctuations.
- Electrical Charge Advantage
- The negative charge of ethosomes allows better attraction to skin surfaces, improving permeability.
- Non-Invasive Delivery System
- Supports active ingredient delivery without the need for invasive techniques—perfect for microneedling, MTS, sonophoresis, or mesotherapy.
- Clean, Skin-Compatible Base
- Free from harsh solvents or harmful emulsifiers, EthosomePTT uses a water-based, dermatologically balanced vehicle.
Ingredient Transparency
(Selected from full list of INCI)
KEY ACTIVES (BRIGHTENING) KEY ACTIVES (ANTI-AC) Niacinamide Niacinamide Tranexamic Acid Tranexamic Acid Ascorbic Acid Salicylic Acid Glutathione Centella Asiatica Extract Ethyl Ascorbyl Ether Colloidal Gold and Platinum
-

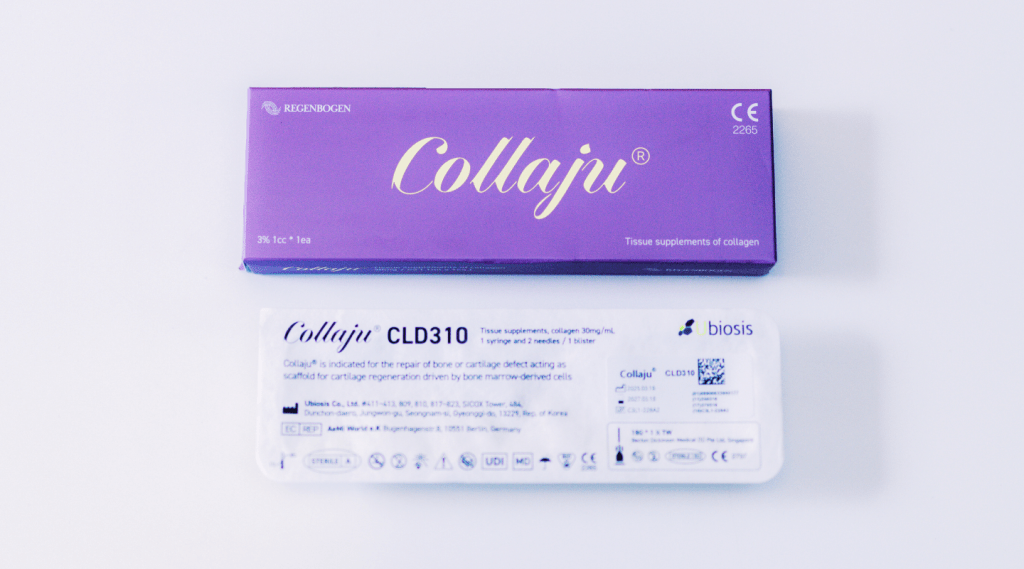
Purified Injectable Atelocollagen for Tissue Regeneration
Collaju is a high-purity, injectable atelocollagen tissue supplement, developed through advanced biotechnology and purified via a patented Aseptic Salt Precipitation Method. This Class 4 medical device is formulated for internal, injectable use and is indicated for both aesthetic rejuvenation and post-procedural tissue support. Each unit contains 1 mL of ready-to-use Type I collagen in liquid form, eliminating the need for mixing or rehydration.
At the molecular level, Collaju retains the triple helix structure of collagen while eliminating immune-reactive telopeptides through enzymatic processing. This atelocollagen structure ensures low antigenicity, high compatibility with human tissue, and rapid integration into the dermal matrix.
Unlike conventional collagen injectables exposed to high heat—which risk denaturation—Collaju is processed under strict aseptic and low-temperature conditions, preserving its native form and function. The resulting collagen fibers are thicker, more stable, and highly bioactive, mirroring the body’s own extracellular matrix and promoting long-term regenerative outcomes.
Scientific Innovation: Salt Precipitation Method
What truly sets Collaju apart is its proprietary salt precipitation process, a refined technique that stabilizes collagen molecules without exposure to high heat or chemical denaturants. This allows for:
- Thicker collagen fiber formation
- Greater resistance to enzymatic degradation
- Higher retention within injected sites
- Reduced inflammatory response
This process results in a product that closely resembles native human collagen in structure and performance, and has been validated through pre-clinical and clinical research for safety and efficacy.
APPLICATIONS
Collaju is ideal for:
- Facial contour restoration and wrinkle reduction
- Acne scar revision and skin texture improvement
- Post-surgical soft tissue reconstruction
- Burn and trauma recovery
- Adjunct in regenerative procedures involving bio-stimulatory agents
Its flexibility across medical and aesthetic indications makes it a valuable tool in dermatology, plastic surgery, and integrative medicine.
MANUFACTURING AND REGULATORY CREDENTIALS
- Product Name: Collaju
- Category: Collagen Tissue Supplement
- Packaging: 1ml/syringe × 1EA per box
- Manufacturer: Ubiosis
- Seller: Juventa Healthcare Co., Ltd.
- Address: 18th Floor, 123 Digital-ro 26-gil, Guro-gu, Seoul
- Storage: Store at room temperature (below 30°C)
- Shelf Life: 2 years from date of manufacture
- Certifications
- KFDA (Korea Food & Drug Administration)
- European CE Certified
- Licensed by NMPA China (National Medical Products Administration)
- ISO-compliant production
Collaju is exclusively distributed in the Philippines by Pascuala Distribution Limited Corporation, the sole authorized distributor of Collaju nationwide. Trusted by clinics and medical institutions alike, Pascuala remains committed to delivering clinically tested and internationally certified medical innovations to Filipino practitioners and patients.
-

Deoxycholic Acid Lipolysis & Adipolysis Solution
EDGE is an advanced injectable solution indicated for the non-surgical reduction of localized fat deposits through adipolysis and lipolysis. It utilizes Deoxycholic Acid in combination with natural botanical compounds to break down fat cell membranes, enhance lipid metabolism, and promote lymphatic clearance. Clinically favored in body contouring, particularly for areas resistant to diet and exercise.
Mechanism of Action
EDGE induces fat cell destruction via a cytolytic mechanism:
- Deoxycholic Acid disrupts adipocyte membranes, leading to irreversible cell lysis. The released lipids are metabolized and cleared through the body’s natural excretory pathways.
- Lipolysis: Triglycerides within the adipocytes are hydrolyzed into glycerol and free fatty acids.
- Adipolysis: Promotes the breakdown of stored fat through metabolic stimulation.
- Adenosine Triphosphate (ATP) supports cellular energy dynamics, facilitating tissue repair, lymphatic activation, and sustained metabolic function during and after the lipolytic process.
Key Bioactive & Natural Ingredients
Formulated with both synthetic and natural components to optimize efficacy and reduce inflammation:
INGREDIENT FUNCTION Deoxycholic Acid Primary fat-dissolving agent; disrupts fat cells Juglans Regia (Walnut) Seed Extract Anti-inflammatory, antioxidant Fucus Vesiculosus Extract Iodine-rich seaweed; enhances circulation and skin elasticity Tyrosine Amino acid precursor; regulates metabolism Aesculus Hippocastanum (Horse Chestnut) Extract Reduces edema, improves venous return Methylpropanediol Solvent and penetration enhancer Methylsilanol Mannuronate Derived from brown algae; promotes collagen, skin firmness Sodium Chloride Osmoregulation and fluid balance FORMULATION PROFILE
- Sterile, water-based injectable solution
- Stabilized with sodium and phosphate derivatives to ensure pH balance
- Enhanced with ATP complexes for improved cellular response
- No added preservatives; formulated for safety and bioavailability
Clinical Use & Target Market
- Exclusively for licensed medical professionals (dermatologists, cosmetic physicians, aesthetic injectors)
- Ideal for localized fat reduction in areas such as:
- Submental region (double chin)
- Upper arms
- Abdomen
- Flanks (love handles)
- Thighs
- Non-surgical alternative to liposuction and other invasive fat-reduction techniques
- Marketed globally under export-only licensing, compliant with professional use restrictions
Medical Disclaimer
EDGE is classified as a cosmeceutical injectable and must only be administered by trained professionals in a clinical setting. Results depend on injection technique, dosage precision, patient selection, and proper post-treatment care. Not intended for use in large-volume fat reduction or in patients with metabolic or hepatic disorders without medical clearance.